India accounts for 26% of the global TB burden, yet drug-induced hepatotoxicity affects up to 28% of patients on standard ATT. Precision monitoring closes the gap between fixed dosing and safe, effective therapy.
Why ATT-TDM?
Standard Dosing
Is Not Enough
Tuberculosis treatment relies on a fixed weight-based regimen, but patient response varies dramatically. Genetics, nutritional status, organ function, HIV co-infection, and concurrent medications all drive unpredictable drug exposures that standard dosing simply cannot account for.
Up to 40% of TB patients have subtherapeutic peak drug concentrations on standard regimens, a leading contributor to acquired drug resistance, prolonged infectiousness, and treatment failure.
At Advait Theragnostics, our ClinPeak™ ATT assay measures actual drug levels in patient plasma, giving clinicians the data they need to dose with confidence and act on toxicity before it becomes irreversible.

ClinPeak™ Assay Configurations
Choose Your Panel
Two validated configurations are designed for different clinical scenarios, from routine monitoring to intensive hepatotoxicity workup.
Single Analytical Workflow
Screening + Quantitation of All 5 First-Line ATT Drugs
One Sample, One Run
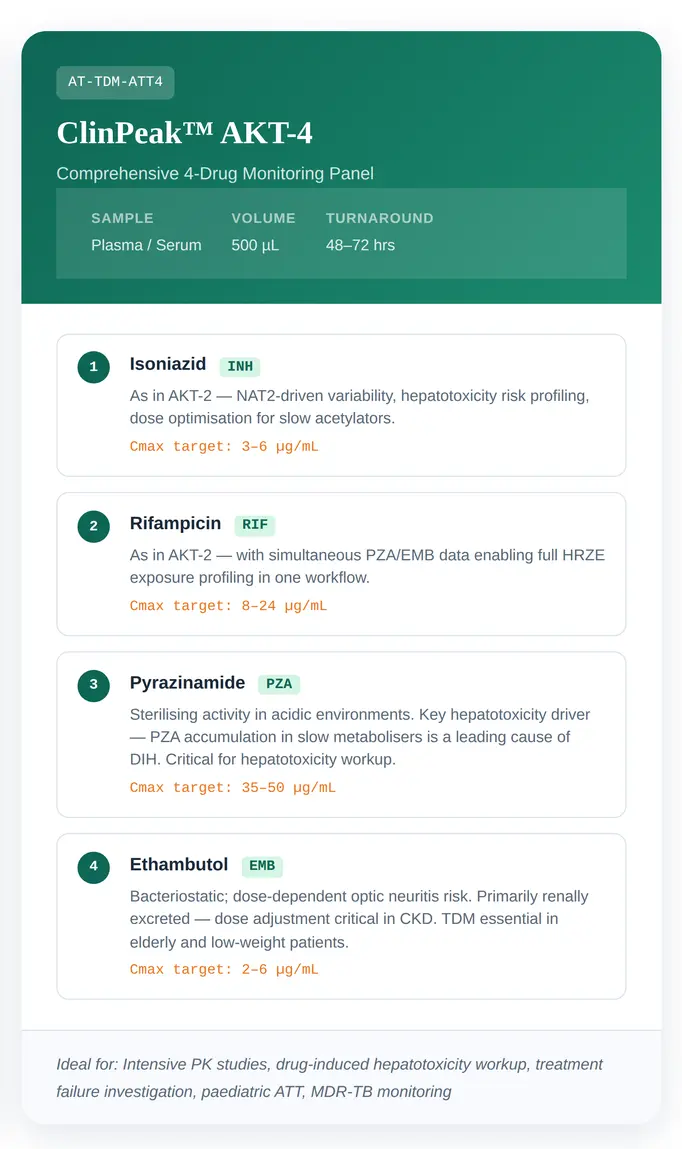
Our full ATT workflow extends to simultaneous detection of all five first-line agents, enabling rapid screening alongside precise quantitation for comprehensive clinical decision support.
*We will soon incorporate the updates in accordance with the New TB Treatment Guidelines 2025 as issued by the National TB Elimination Programme (NTEP), Government of India.
Hepatotoxicity Correlation
Powered by METLINE 2.0™
When liver enzymes rise, METLINE 2.0™ connects the dots, correlating ATT drug levels with clinical biomarkers to identify the culprit drug, guide safe therapy interruption, and plan evidence-based re-challenge
1
Drug–Liver Biomarker Correlation
2
Personalised Dose Optimisation
3
Temporal Trend Analysis
4
Safe Re-challenge Guidance
Test Directory
Ordering Information
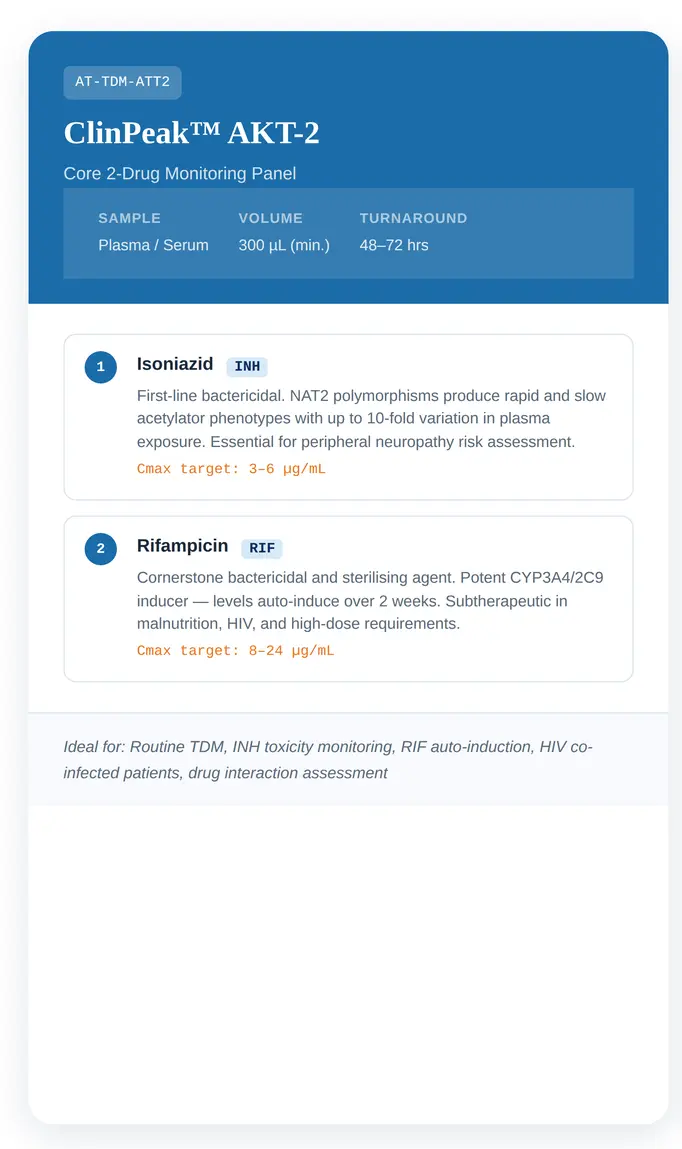
AT-TDM-ATT2
- Plasma / Serum
- 300 µL
- 48–72 hrs
- Isoniazid, Rifampicin
AT-TDM-ATT5
- Plasma / Serum
- 600 µL
- 48–72 hrs
- INH, RIF, PZA, EMB, Streptomycin
AT-TDM-DIH
- With LFT requisition
- -
- 72-96 hrs
- Add-on to AKT-4 or ATT5
Get Started
Right Drug. Right Dose. Right Time.
Connect with our clinical specialists to discuss ATT-TDM protocols, panel selection, or METLINE 2.0™ integration for your institution.
